 
The metal had two small diversions (slits), leading to an electrometer that could measure a small. He built his cathode ray tube with a metal cylinder on the other end. The charge-to-mass ratio could then be calculated, and was found to be at least 1,000 times higher than that of the hydrogen atom. Thompson, conducted his first cathode ray tube experiment to prove that rays emitted from an electron gun are inseparable from the latent charge. An equal deflection was produced by applying a magnetic field, perpendicular to the electric field, using an electromagnet. The beam was deflected by passing it through an electric field between the parallel plates. In the apparatus shown here, rays produced at the cathode were focused into a narrow beam by the slits in the cylinders in the narrow part of the tube, producing a fluorescent spot on the spherical end of the tube. However, when atoms were studied in large electric fields, evidences came up indicating that they consisted of subatomic particles. The Cathode ray experiment is based on conduction of electricity through gases at low pressure in a discharge tube. Thomson admitted that when he first performed the experiment he. Cathode rays are deflected by both magnetic and electric fields. He concluded that the electrostatic properties of the cathode ray are either nil or very feeble. By 1897, Thomson had measured the charge-to-mass ratio, but finding the charge or mass of the electron independently took a further two years. The cathode ray discharge tube experiment does not lead to the discovery of radioactivity. 
Others, such as geophysicist Emil Wiechert, realized that the charged carriers of electricity were very small, but Thomson seems to have been the first to suggest that electrons - or 'corpuscles', as he called them - existed inside the atom he put forward a model for atomic structure that was based on the patterns of repulsion of floating magnets noted by Alfred Mayer in 1878. This earlier generation had observed that these rays travelled in straight lines but could be deflected by a magnetic field, which became the basis of Thomson's work. He demonstrated that cathode rays were negatively. The discharge tubes and pumps invented by Heinrich Geissler in the 1850s, and the induction coil perfected by Heinrich Ruhmkorff at about the same time, had enabled many physicists, such as Julius Plücker and Johann Hittorf, to experiment with the new 'cathode rays'. Thomson discovered the electron by experimenting with a Crookes, or cathode ray, tube. The road leading to the discovery had been an arduous one, trodden by many scientists. Although it was certainly one of his instruments, he used a series of cathode-ray tubes in that year, so we cannot be certain.Īlthough the moment of discovery of the first subatomic particle has been controversial, Thomson's measurement of the charge-to-mass ratio for cathode rays is now considered to have defined the electron. 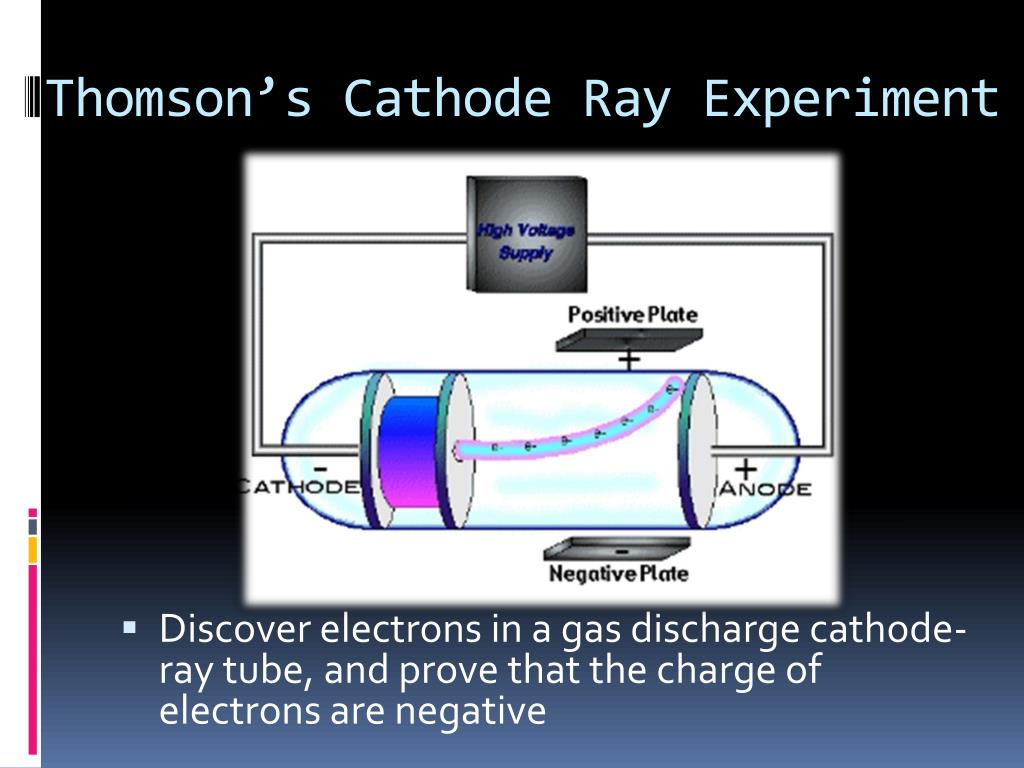
This has been claimed to be the apparatus with which Joseph John Thomson discovered the electron in 1897. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
